N-Boryl pyridyl anion chemistry
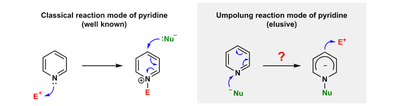
As a representative N-heterocycle, pyridine exhibits a common reaction mode, in which the N atom attacks an electrophile and a nucleophile could add to the activated pyridine ring. In contrary, a polarity-inversed reaction mode, in which a nucleophile attacks the N-site of pyridine to form an N-substituted nucleophilic pyridine anion, remained elusive before our study.
In an early study, we discovered that pyridine derivatives could catalyze the transition-metal-free borylation reaction of aryl halides with diboron(4) compounds. Detailed mechanistic study revealed an unprecedented reaction mode of pyridine and diboron(4) in the presence of an alcoholate to generate a novel N-boryl pyridyl anion key intermediate, which mimics a nucleophilic addition of a boryl anion to the N-site of pyridine to form a Meisenheimer-type anion.
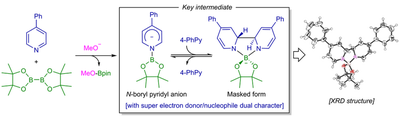
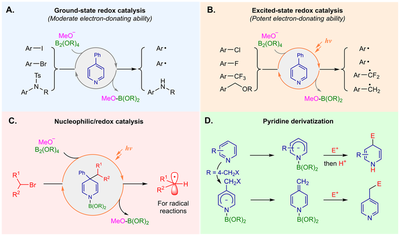
These discoveries constitute an important addition to the known reaction mode of pyridine, and demonstrate a new organocatalytic mode of pyridine.
Major contributors
Key references
- Zhang, Li; Jiao, Lei* “Pyridine-catalyzed radical borylation of aryl halides.” J. Am. Chem. Soc. 2017, 139, 607-610.
- Zhang, Li; Jiao, Lei* “Super electron donors derived from diboron.” Chem. Sci. 2018, 9, 2711-2722.
- Zhang, Li; Jiao, Lei* “Visible-light-induced organocatalytic borylation of aryl chlorides.” J. Am. Chem. Soc. 2019, 141, 9124-9128.
- Yang, Huan; Zhang, Li; Zhou, Fei-Yu; Jiao, Lei* “An umpolung approach to the hydroboration of pyridines: a novel and efficient synthesis of N-H 1,4-dihydropyridines.” Chem. Sci. 2020, 11, 742-747.
- Zhang, Li; Wu, Zhong-Qian; Jiao, Lei* “Photoinduced radical borylation of alkyl bromides catalyzed by 4-phenylpyridine.” Angew. Chem. Int. Ed. 2020, 59, 2095-2099.
- Zhou, Fei-Yu; Jiao, Lei* “Asymmetric defluoroallylation of 4-trifluoromethylpyridines enabled by umpolung C-F bond activation.” Angew. Chem. Int. Ed. 2022, 61, e202201102.
- Bai, Lutao; Jiao, Lei* “Modular photoredox system with extreme reduction potentials based on pyridine catalysis.” Chem 2023, 9, 3245-3267.
- Zhou, Fei-Yu; Jiao, Lei* “4-Pyridyl perfluoroalkyl sulfide as a practical nucleophilic perfluoroalkylation reagent.” Chem 2024, 10, 2311-2323.
- Zhang, Li; Jiao, Lei* “N-Boryl pyridyl anion chemistry.” Acc. Chem. Res. 2025, 58, 1023-1035.