Construction of Indoline/Indolenine Ring Systems by a Palladium-Catalyzed Intramolecular Dearomative Heck Reaction and the Subsequent Aza-Semipinacol Rearrangement.
Abstract
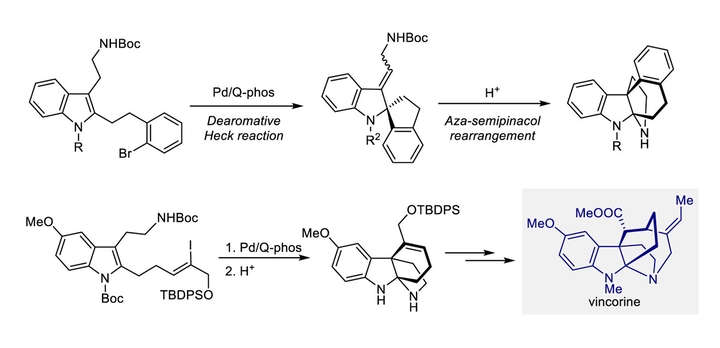
The palladium-catalyzed intramolecular dearomative Heck reaction of 2,3-disubstituted indoles serves as an access to spiro-indoline products. Herein, we report an efficient construction of indoline/indolenine core stuctures via a dearomative Heck reaction of simple 2,3-disubstituted indoles with all-carbon tethers and the subsequent aza-semipinacol rearrangement. The Heck reaction features a high C2-selectivity, and the stereospecific aryl/alkyl migration selectivity has been investigated by DFT calculations. Using this method, we accomplished the formal total synthesis of akuammiline alkaloids vincorine.
Publication
J. Org. Chem. 2021, 86, 5727-5743.