Abstract
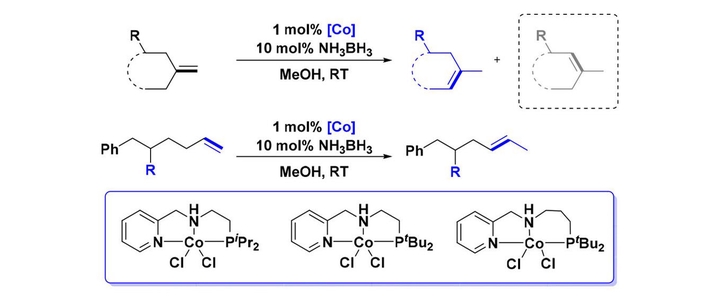
Olefin isomerization is a significant transformation in organic synthesis, which provides a convenient synthetic route for internal olefins and remote functionalization processes. The selectivity of an olefin isomerization process is often thermodynamically controlled. Thus, to achieve selectivity under kinetic control is very challenging. Herein, we report a novel cobalt-catalyzed regioselective olefin isomerization reaction. By taking the advantage of fine-tunable NNP-pincer ligand structures, this catalytic system features high kinetic control of regioselectivity. This mild catalytic system enables the isomerization of 1,1-disubstituted olefins bearing a wide range of functional groups in excellent yields and regioselectivity. The synthetic utility of this transformation was highlighted by the highly selective preparation of a key intermediate for the total synthesis of minfiensine. Moreover, a new strategy was developed to realize the selective monoisomerization of 1-alkenes to 2-alkenes dictated by installing substituents on the $γ$-position of the double bonds. Mechanistic studies supported that the in situ generated Co– H species underwent migratory insertion of double bond/$β$-H elimination sequence to afford the isomerization product. The less hindered olefin products were always preferred in this cobalt-catalyzed olefin isomerization due to an effective ligand control of the regioselectivity for the $β$-H elimination step.